|
emLas® vs. MicroLight® ML830 |
 |
MicroLight Corporation of
American |
| |
is the first one received FDA clearance
to market low-level laser therapy for the non-surgical
treatment of Carpal Tunnel Syndrome. (Please refer MicroLight
web site  ) ) |
|
 |
Konftec Corporation |
| |
The taping laser released from Konftec Corporation
had got appreciations from master experts since the beginning
of 2005: |
| |
˙" A great step in LLLT ---- " |
| |
˙" Revolutionary invention ---- " |
| |
˙" Make LLLT more acceptable ---- " |
|
| Manufacturer |
Konftec |
MicroLight
|
Model
No.
(Click for Web Page) |
emLas-150 |
emLas-650 |
ML830 |
| Reference Picture |
 |
 |

|
| Price |
| Market
Price |
USD 840 |
USD 1,560 |
ML830 DC: USD
3,995
ML830 Battery: USD 4,995 |
| Laser
Part |
| Laser Wavelength |
780nm
808nm |
780nm
808nm |
830nm |
| Quantity
of Laser |
5 |
3 |
| Laser Output Power |
250mW (5 x 50mW) |
780nm: 500mW (5 x 100mW)
808nm: 700mW (5 x 140mW) |
90mW (3 x 30mW) |
| Laser
Penetrate Depth |
3 ~ 5 cm |
2 inch (5 cm):
As manufacturer's specification |
| Operation |
| Treatment
Method |
Wrap;
5 sets of laser operate
simultaneously |
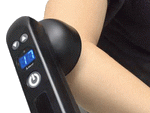
Handheld;
move to treat step-by-step |
Handheld;
move to treat step-by-step |
 |
 |
 |
| Accurate Laser Dose |
 Time
Control Time
Control |
 |
| Process Indication |
 LED
dynamic display LED
dynamic display
|
 |
| Auto Shut-off |
 with
beep alert with
beep alert |
 |
| Safety
& Verification |
| Necessity of
Wearing Protection goggles |
 |
 Patient & Practitioner Patient & Practitioner |
 Patient & Practitioner Patient & Practitioner |
Quality Certification
|
ISO
13485: 2003 |
 |
 Verification Verification
|
CE Verification of Compliance:
EMC Directive 93/42/EEC
(Operation Environment:
Medical Electrical Equipment)
Standard Applied: IEC 60601-1 and EC 60601-1-2 |
 |







